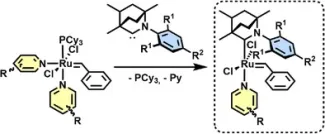
Grubbs-type ruthenium–alkylidene catalysts are pivotal in olefin metathesis, with improved activity and initiation achieved through ligand design. While NHC- and CAAC-based systems are well established, robust and strongly σ-donating BICAACs remain underexplored, particularly in pyridine-activated third-generation catalysts important for Ring-Opening Metathesis Polymerization (ROMP). Here we report the first synthesis and full characterization of pyridine-ligated BICAAC–Ru alkylidene complexes. Using a modified Grubbs protocol that avoids bis-carbene formation, pyridine complexes (1a–3a, 3b) were prepared from in situ generated BICAACs and bis-pyridine phosphine intermediates. Spectroscopic, crystallographic, and DFT studies show that steric congestion at Ru disfavors bis-pyridine coordination, with a second pyridine. These results establish pyridine-coordinated BICAAC–Ru complexes as a well-defined platform for next-generation metathesis catalysts.
Graphical Abstract
First synthesis and comprehensive characterization of pyridine-ligated BICAAC–Ru benzylidene complexes. The compounds were obtained using a modified Grubbs protocol that prevents bis-carbene formation, starting from in situ generated BICAAC ligands and bis-pyridine ruthenium phosphine intermediates.